Biotech Company Financial Model
This 20-Year, 3-Statement Excel Biotech Company Financial Model includes revenue streams from Biopharmaceutical Drug sales, Contract Manufacturing, Royalty Revenue, etc. Cost structures and financial statements, Discounted Cash Flow (DCF) with Terminal Value, Sensitivity Analysis, and WACC to forecast the financial health of your Biotech company. Click here for a Synthetic Biology Model.
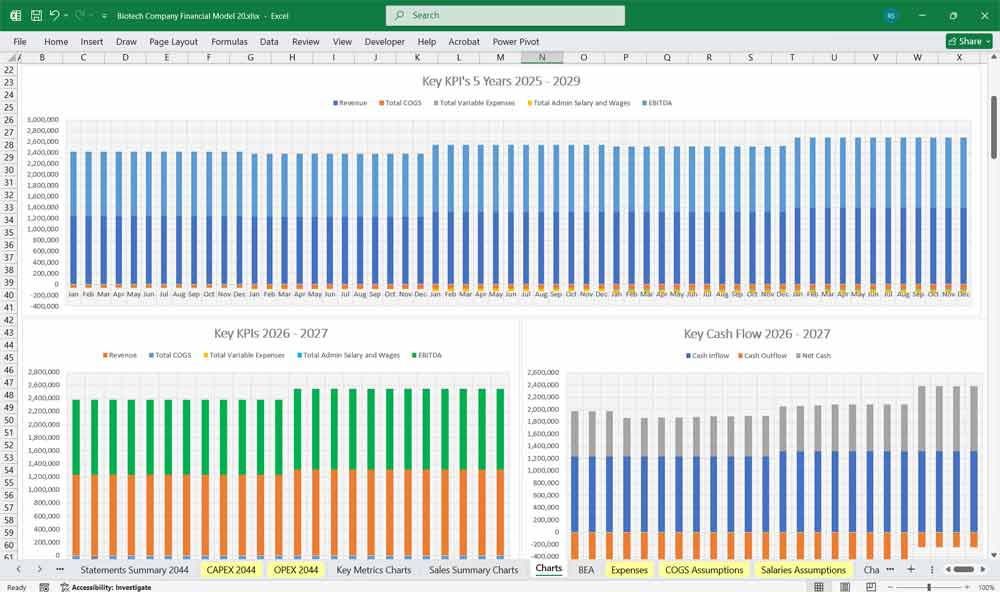
20-year Financial Model For a Biotech Company
This very extensive 20-Year Biotech Company Model involves detailed revenue projections, cost structures, capital expenditures, and financing needs. This model provides a thorough understanding of the financial viability, profitability, and cash flow position of the centre. Including: 20x Income Statements, Cash Flow Statements, Balance Sheets, CAPEX sheets, OPEX Sheets, Statement Summary Sheets, and Revenue Forecasting Charts with the specified editable revenue streams, BEA charts, sales summary charts, employee salary tabs and expenses sheets.
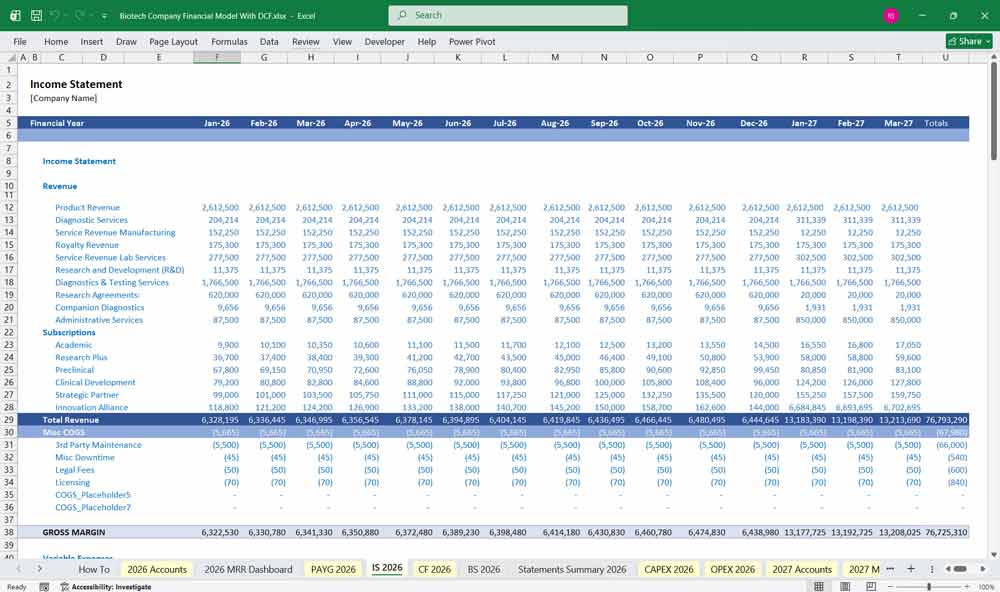
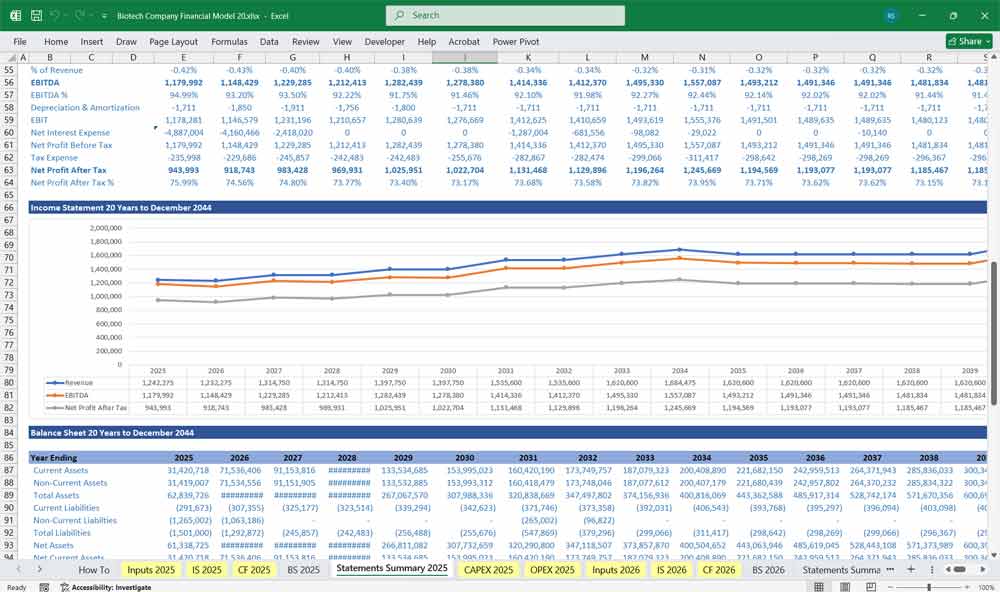
Income Statement (Profit & Loss Statement)
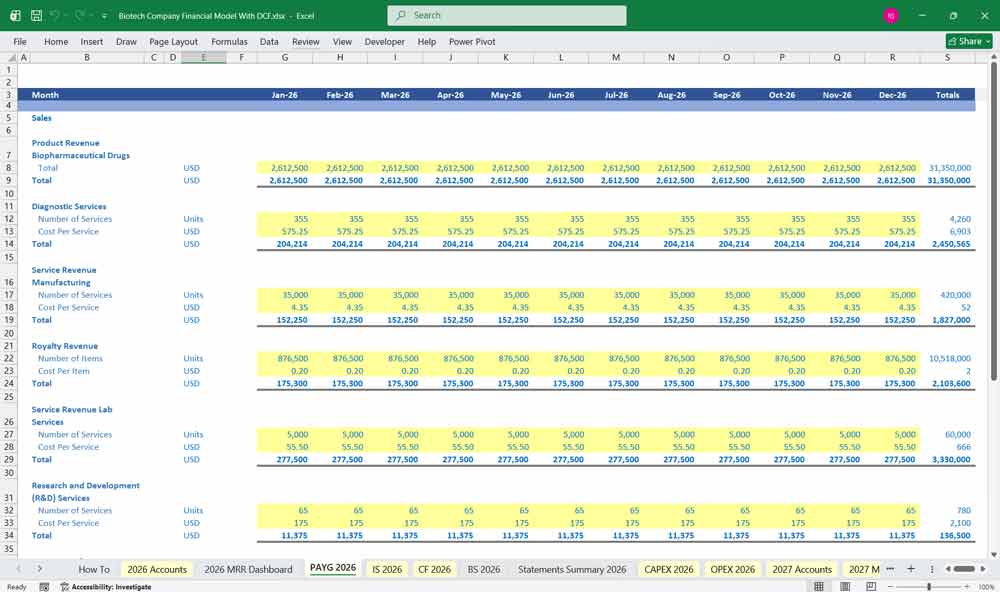
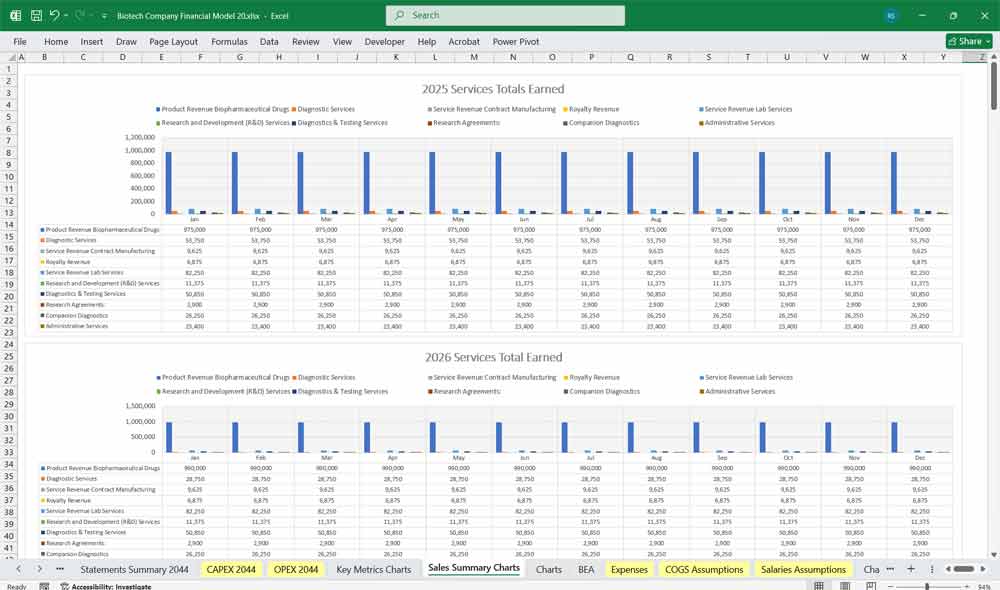
Revenue Streams
Biopharmaceutical Drug Sales
Revenue from proprietary drug sales (post-approval).
Segmented by product, geography, or therapeutic area.
Key drivers: price per unit, volume (patients), market share, and penetration rate.
Diagnostic Services
Clinical diagnostics tests offered to hospitals or physicians.
Revenue modeled by number of tests × price/test.
Contract Manufacturing Revenue
Revenue from manufacturing biologics or small molecules for third parties.
Modeled by volume (batches produced) and unit pricing.
Royalty Revenue
Based on agreements with partners using patented tech or licensed drugs.
Typically a percentage of net sales (e.g., 5–12%).
Service Revenue (Lab Services)
Analytical testing, biomarker analysis, clinical trial support.
Project-based or volume-based billing.
BIO R&D Revenue
Sponsored R&D contracts or grants (e.g., NIH, DARPA).
Recognized over time or at milestones.
Diagnostics & Testing Services
Includes proprietary testing platforms (e.g., PCR, genomics).
Revenue per test or bundled diagnostics packages.
Cost of Goods Sold (COGS)
Segmented by revenue line (e.g., cost to manufacture drugs, diagnostic reagents).
Includes:
Raw materials
Lab consumables
Personnel directly involved in production
Facility and depreciation costs
Gross Profit
Revenue – COGS
Gross Margin per revenue stream is useful for assessing profitability.
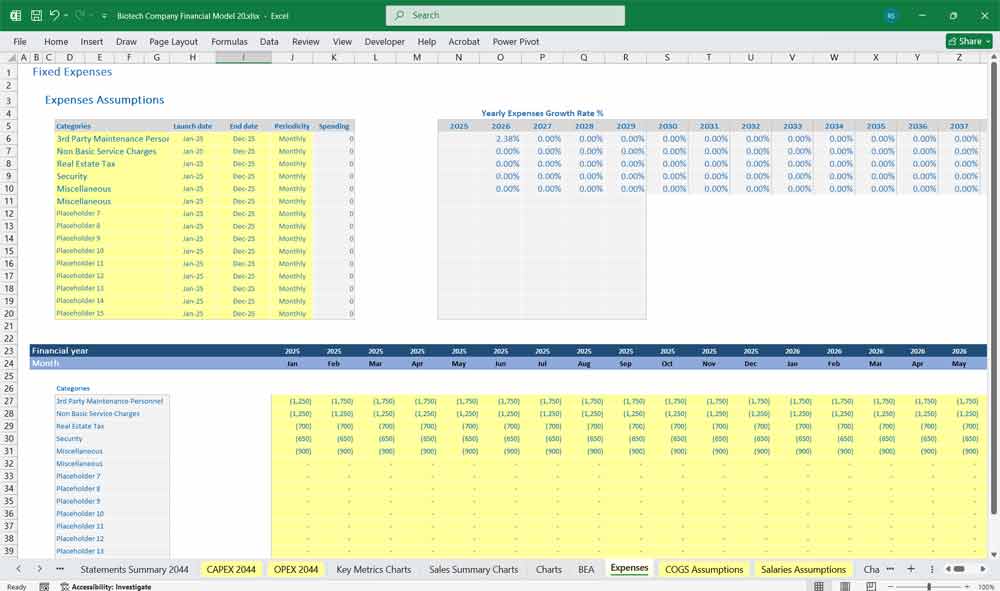
Operating Expenses
Research & Development (R&D)
Preclinical studies, clinical trials (Phases I–III), regulatory submission.
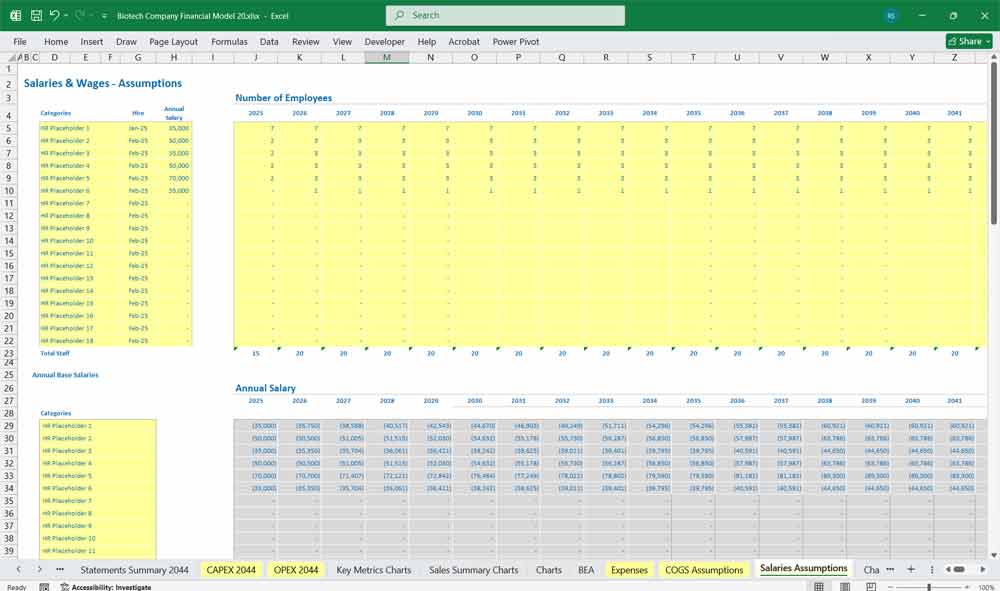
Salaries for scientists, CRO payments, lab costs.
Capitalized or expensed depending on stage.
Selling, General & Administrative (SG&A)
Commercial team salaries, marketing, legal, finance, HR, office space.
Depreciation & Amortization
From lab equipment, facilities, capitalized IP or licenses.
Other Operating Expenses
Stock-based compensation
IP litigation, licensing fees
Operating Income (EBIT)
Gross Profit – Operating Expenses
Interest and Taxes
Interest Expense: From biotech debt or venture loans.
Interest Income: Cash investments, if applicable.
Tax Expense: May be zero in early years due to losses/NOLs.
Net Income
EBIT – Interest + Other Income – Taxes
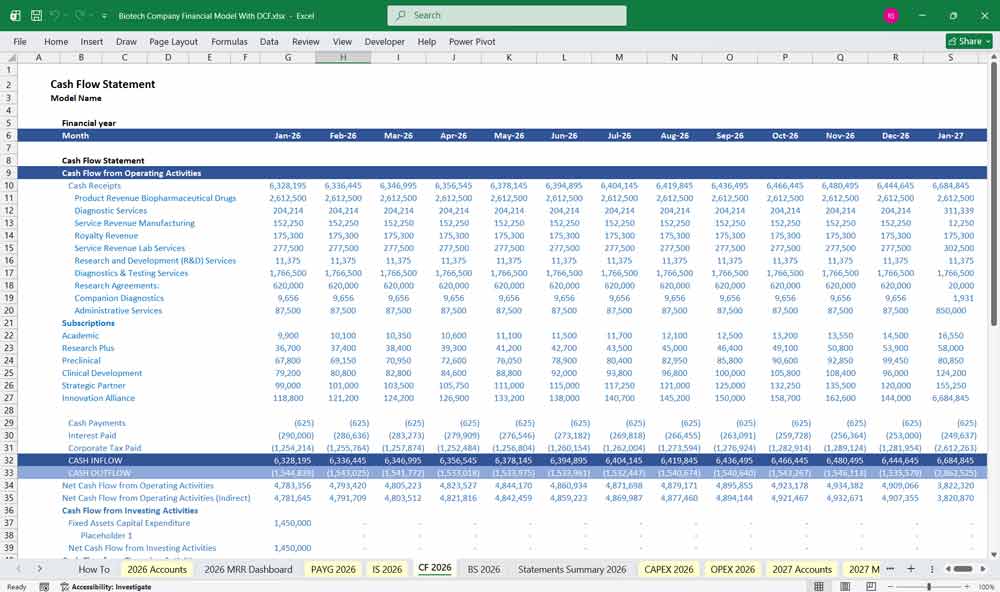
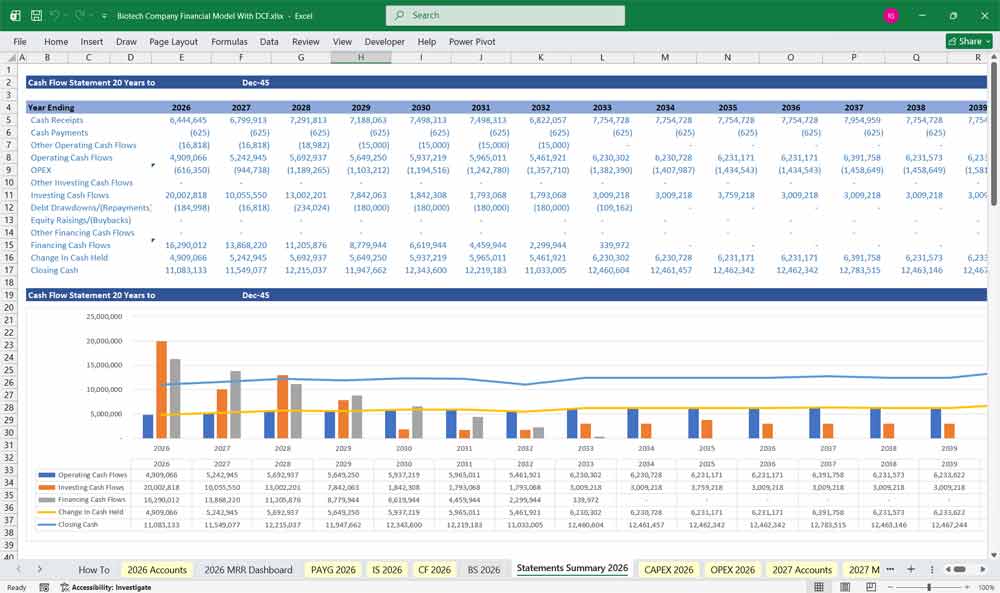
Biotech Company Cash Flow Statement
Cash Flow from Operating Activities
Net Income
Adjustments:
Depreciation & amortization
Stock-based compensation
Deferred taxes
Changes in Working Capital:
Accounts receivable
Inventory (especially for diagnostic kits, drug stock)
Accounts payable
Prepaid expenses
Accrued liabilities
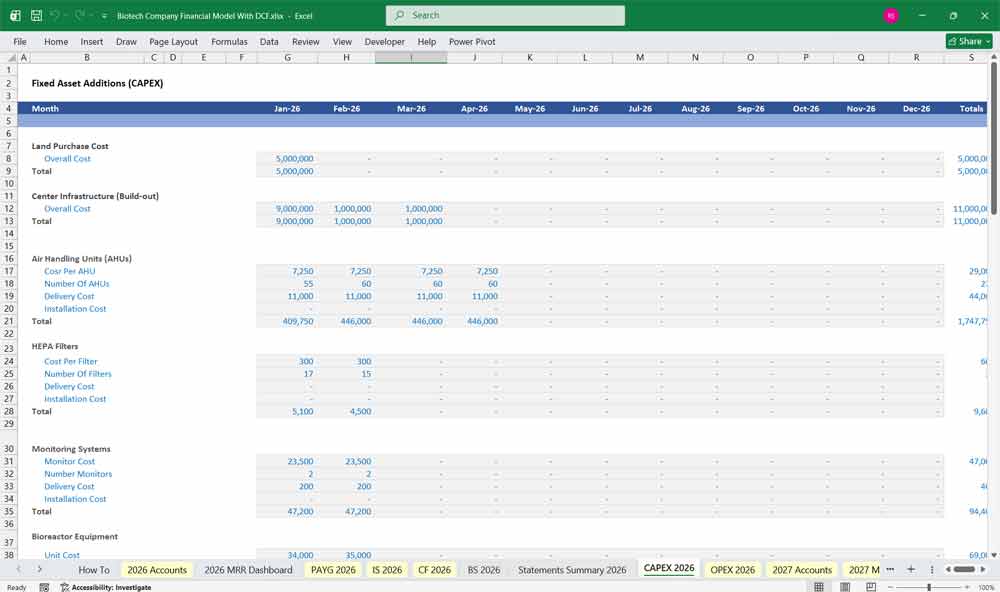
Cash Flow from Investing Activities
Capital Expenditures (CapEx):
Lab equipment, diagnostics machinery, facilities.
Acquisition of IP or Licenses
Investment in Joint Ventures or Affiliates
Purchases/Sales of Marketable Securities
Cash Flow from Financing Activities
Issuance of Common Stock / Equity Raises
IPO, follow-on offerings, private placements.
Debt Issuance or Repayment
Interest Payments
Warrants/Options Exercises
Government Grants or Non-Dilutive Funding
Net Change in Cash
Ending Cash = Beginning Cash + Net Cash from Operating + Investing + Financing
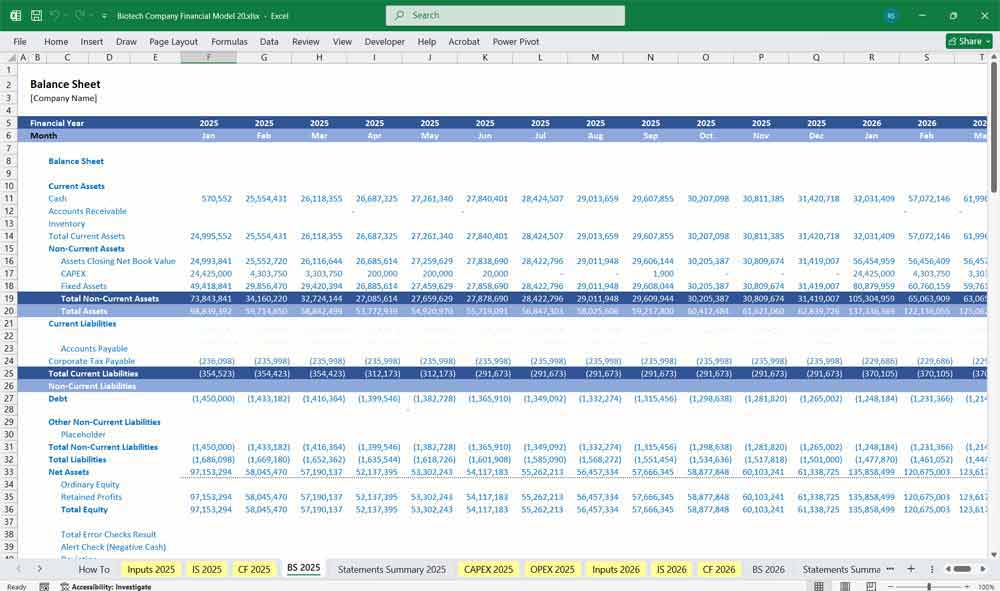
Biotech Company Balance Sheet
Assets
Current Assets
Cash and Cash Equivalents
Marketable Securities
Accounts Receivable
Inventory: Raw materials, WIP, finished goods (for drugs or kits).
Prepaid Expenses and Other Current Assets
Non-Current Assets
Property, Plant, Equipment (PP&E): Labs, bioreactors, testing machines.
Intangible Assets: Patents, licenses, acquired IP.
Deferred Tax Assets: From net operating losses.
Goodwill: If acquisitions occurred.
Liabilities
Current Liabilities
Accounts Payable
Accrued Liabilities: CRO payments, R&D trials, payroll.
Deferred Revenue: Upfront from R&D contracts or licensing deals.
Current Portion of Debt
Long-Term Liabilities
Convertible Debt / Venture Loans
Deferred Tax Liabilities
Long-Term Lease Liabilities
Key Financial Metrics for a Biotech Company
Revenue Metrics
Biotech Company Biopharmaceutical Drug Sales
Biotech companies generate significant revenue from the sale of proprietary biopharmaceutical drugs developed through in-house R&D. These drugs often target complex or rare diseases and command premium pricing due to their innovation and therapeutic value. Revenue from drug sales typically becomes substantial following regulatory approval (e.g., FDA, EMA) and commercialization, and is a primary driver of long-term profitability. Revenue forecasting includes pricing, market penetration, patient population, and reimbursement assumptions.
Diagnostic Services from a Biotech Company
Diagnostic services are a growing revenue stream for biotech companies involved in molecular or clinical diagnostics, and these services may include genetic testing, disease screening, or biomarker detection for hospitals, clinics, or research institutions. Revenue is generally based on the number of tests performed multiplied by the fee per test. Diagnostic offerings can be proprietary platforms or standardized assays and often provide recurring revenue once adopted in clinical workflows.
Contract Manufacturing and a Biotech Company
Biotech companies with advanced manufacturing capabilities may offer contract manufacturing services to third-party pharmaceutical or biotech firms, and this includes the production of biologics, small molecules, or cell and gene therapies under cGMP conditions. Revenue is typically derived from volume-based agreements or long-term manufacturing contracts. Contract manufacturing leverages excess capacity and infrastructure to generate additional, stable income while diversifying the business model.
Royalty Revenue Gains For A Biotech Company
Royalty revenue for a biotech company stems from licensing proprietary technologies or drug candidates to partners, who then commercialize the products. The biotech company earns a percentage of the partner’s net sales, often ranging from 5% to 12%. This non-operational income can be significant, especially when the licensed product achieves global commercial success. Royalties provide high-margin, recurring revenue without ongoing operational cost burdens.
Service Revenue And Biotech Company
(Lab Services)
Biotech companies frequently offer lab services such as biomarker analysis, assay development, or clinical trial support. These services are marketed to pharmaceutical companies, academic institutions, or contract research organizations (CROs). Service revenue is generally project-based or fee-for-service and can scale with demand for outsourced R&D. This stream helps monetize excess lab capacity and strengthens relationships with potential partners.
BIO R&D Revenues For A Biotech Company
Some biotech companies receive R&D revenue through partnerships, government grants, or collaborative research agreements. This revenue supports early-stage development and is typically recognized as milestones are achieved or work is performed. Examples include NIH or BARDA grants, co-development deals, or sponsored preclinical and clinical work. BIO R&D revenue helps offset development costs and reduces reliance on equity financing.
Biotech Company Diagnostics & Testing Services
Biotech companies offering diagnostics and testing services may generate revenue from proprietary platforms such as PCR-based tests, genomic sequencing, or companion diagnostics. These services are provided to healthcare providers, laboratories, or clinical researchers. The revenue is often volume-based and benefits from recurring demand, particularly when tests are integrated into disease management protocols. This segment is essential for companies focused on personalized medicine.
6-Tier Subscription Model Add-On
Biotech firms are increasingly pivoting toward subscription models for “Genomics-as-a-Service” and continuous health monitoring, effectively trading one-off diagnostic fees for the high-margin stability of recurring revenue.
Tier 1 – Explorer (Academic & Early Research Access)
Target Audience: Individual researchers, graduate labs, early-stage biotech startups
Purpose: Low-cost entry for early research and hypothesis generation
Key Features:
Limited access to curated biological datasets (e.g., genomics, proteomics, or assay data)
Basic bioinformatics analysis tools (cloud-based)
Standard documentation and research-use-only (RUO) license
Community forum support
Monthly data updates
Download limits and capped compute hours
Value Proposition:
Affordable access to high-quality biological data and tools to accelerate early discovery without heavy infrastructure investment.
Tier 2 – Research Plus (Advanced Lab Access)
Target Audience: University departments, small biotech R&D teams
Expanded Capabilities:
Full dataset library access (including historical data archives)
Advanced analytics (AI-assisted target identification, pathway modeling)
Increased compute capacity
API access for lab automation integration
Priority email support
Quarterly expert webinars and training sessions
Value Proposition:
Accelerates discovery pipelines through broader datasets and deeper computational capabilities.
Tier 3 – Translational (Preclinical Development Support)
Target Audience: Venture-backed biotech startups, translational medicine teams
Added Benefits:
Integrated multi-omics datasets
Predictive modeling for toxicity & efficacy
Custom biomarker discovery tools
Dedicated customer success manager
Secure collaborative workspaces
Early access to beta platform features
Optional add-on wet-lab validation services
Compliance Level:
Preclinical data standards aligned with regulatory frameworks such as U.S. Food and Drug Administration guidance.
Value Proposition:
Bridges research and IND-enabling studies with regulatory-aligned data and validation tools.
Tier 4 – Clinical Development (Regulatory-Grade Platform)
Target Audience: Mid-size biotech firms entering clinical trials
Enterprise-Grade Features:
GxP-compliant data environment
21 CFR Part 11-compliant digital audit trails
Secure patient data handling (HIPAA-compliant infrastructure)
Clinical trial biomarker stratification tools
Regulatory documentation generation modules
Direct consultation access with regulatory affairs experts
Data hosting options in validated cloud environments (e.g., Amazon Web Services GovCloud)
Value Proposition:
Enables seamless transition from preclinical research into Phase I/II trials with built-in compliance infrastructure.
Tier 5 – Strategic Partner (Enterprise Biopharma Collaboration)
Target Audience: Large pharmaceutical companies & global biotech corporations
Premium Capabilities:
Dedicated private data environments
AI-driven drug target pipeline integration
Custom algorithm development
On-premise or hybrid deployment options
Dedicated scientific advisory team
SLA-backed uptime guarantees (99.9%+)
Integrated real-world evidence data streams
Collaboration Model:
Long-term licensing agreements and co-development revenue-sharing opportunities with strategic oversight from executive stakeholders.
Value Proposition:
Transforms the platform into a scalable R&D engine embedded into enterprise drug discovery workflows.
Tier 6 – Innovation Alliance (Co-Development & IP Partnership)
Target Audience: Global pharma leaders, government agencies, and multinational research consortia
Elite-Level Offerings:
Joint IP ownership frameworks
Fully customized platform architecture
Embedded bioinformatics & data science teams
Direct integration into global regulatory submissions
Advanced AI/ML co-development
Access to global regulatory insights including alignment with the European Medicines Agency
Multi-region data sovereignty solutions
Strategic Structure:
Multi-year agreements
Milestone-based payments
Equity or licensing structures
Co-branded innovation programs
Value Proposition:
Moves beyond subscription — becomes a strategic innovation engine and long-term biotech co-creation partnership.
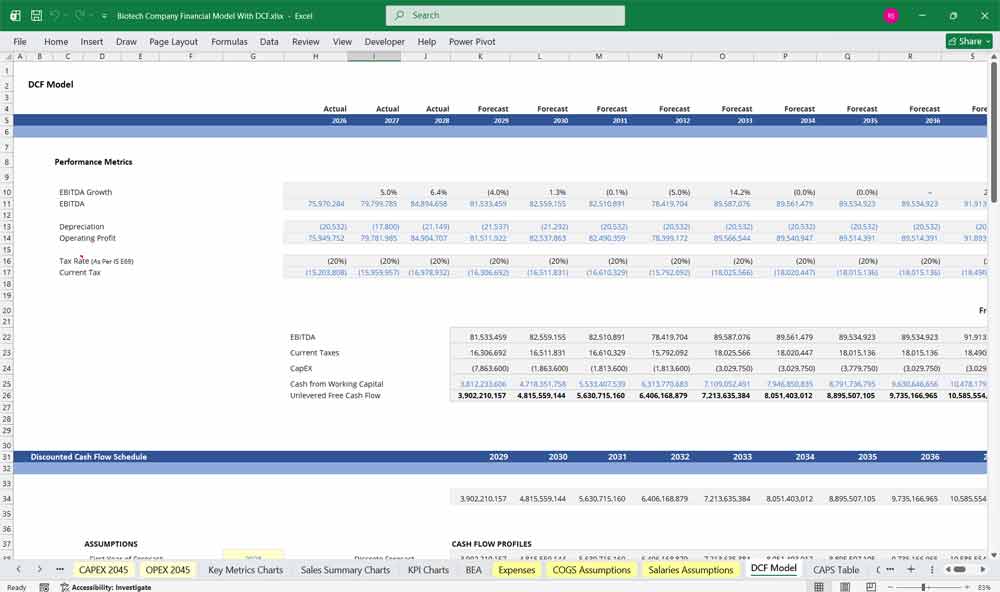
Valuing Your Biotech Company With A Discounted Cash Flow Value (DCF)
Valuing the “Binary” Pipeline
This Discounted Cash Flow (DCF) analysis values a biotech company by projecting its future cash flows—often tied to milestone-based drug development, licensing revenues, or commercialization timelines—and discounting them back to present value. In biotech, DCF models typically incorporate probability-adjusted cash flows to reflect clinical trial risk and regulatory uncertainty from agencies like the U.S. Food and Drug Administration. Because revenues may be years away, especially for pre-revenue firms, assumptions around peak sales, patent life, pricing power, and time to market heavily influence valuation outcomes.
WACC: Pricing Clinical Failure and Venture Risk
WACC represents the blended cost of equity and debt financing used to discount projected cash flows in a DCF model. For biotech companies, WACC is generally higher than in mature industries due to elevated R&D risk, regulatory exposure, and revenue volatility. Early-stage biotech firms often rely primarily on equity financing, increasing the cost of capital due to dilution risk and investor return expectations. As a company advances into later clinical phases or secures partnerships, its perceived risk profile may improve, potentially lowering WACC.
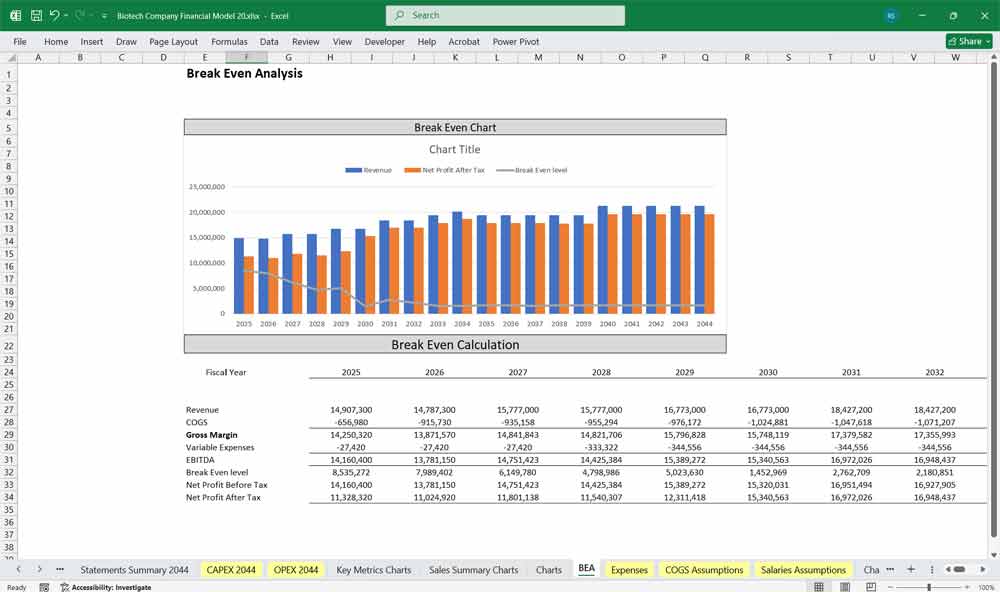
Sensitivity Analysis: Stress-Testing the “Probability of Success”
Sensitivity analysis evaluates how changes in key assumptions impact a biotech company’s valuation. Given the sector’s binary clinical outcomes, analysts often test variables such as probability of trial success, time to regulatory approval, peak market penetration, pricing assumptions, and WACC. Even small adjustments to these inputs can significantly alter valuation results, especially for companies dependent on a single drug candidate. This approach helps investors understand downside risk, upside potential, and which variables most materially drive enterprise value.
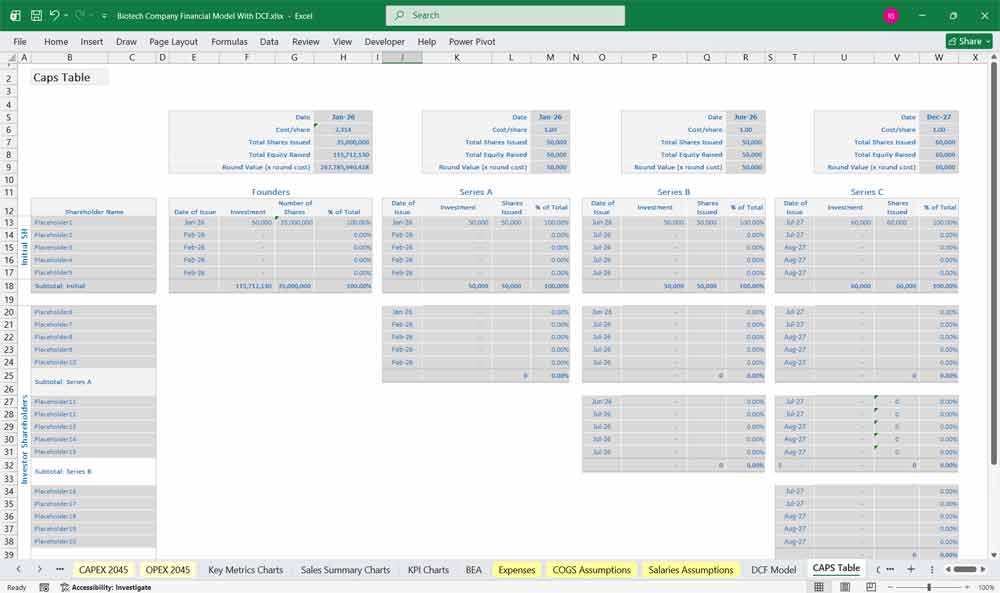
Final Notes on the Financial Model
This 20-Year Biotech Company Financial Model must focus on balancing capital expenditures with steady revenue growth from diversified bio managed services. And so by optimizing operational costs, and power efficiency, and maximizing high-margin services like drug sales and diagnostics, the model ensures sustainable profitability and cash flow stability.
Download Link On Next Page
Download Link On Next Page
